Hypothesis: If Rate of reaction is related to the surface area of the Alka Seltzer, then the more suface area of the tablet exposed the faster the rate of reaction. The crushed tablet will react quickly, while the whole tablet will react slowly.
Independent variable: Suface area of the Alka Seltzer
Dependent variable: Rate of reaction
Controlled variable: amount of Alka Seltzer, How crushed it is, temperature, amount of water.
Materials:
-Mortar and Pestle
-Alka Seltzer Tablets x3 (Sodium Carbonate)
-Water (200ml)
-4 beakers
-knife or saw
-3 pieces of paper
-4 timers
Procedure:
1. Take out the 3 tablets and use the mortar and pestle to crush one of the tablets into a fine powder.
2. Use the saw and cut another one of the tablets into smaller squares.
3. place the powder, tablet square and whole tablet all on different pieces of paper.
4. Take the 4 beakers and place them next to each other number the breakers and the pieces of paper the tablets are on. (the fourth beaker is the control.)
5. Fill the beakers with 40ml of Water
6. Simultaneously pour/place the different states of tablet into each respective breaker
7. Start the timers right after the tablets are poured in, see which one has reacted the fastest
8. Observe the reaction and note the changes and speed of reaction.
9. Clean up and put equipment away.
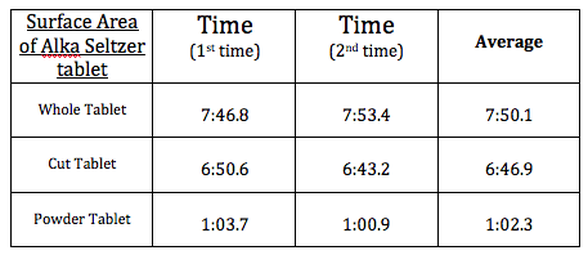
Data table: The rate of which the tablets dissolve.
In my hypothesis I stated that the more surface area exposed the faster the rate of reaction. My hypothesis was proven correct. The crushed tablet had the fastest reaction rate and the whole tablet the slowest. My predictions were all right. I knew this would happen because surface area is a key factor in rate of reaction, when there is more surface area exposed the atoms and molecules have a higher chance of bumping into each other and reacting then with a low surface area.
The procedure and materials were well thought up. Everything the experiment needed was included. The way it was conducted made the result valid and reliable. All variables were thought or, the tablets were all the same size for each category of surface area, the temperature of the water was the same, the amount of water was the same, and the amount of tablet was the same. Nothing could have changed or faulted the resultes of the experiment. The independent variable was the surface area of the tablet and the dependent variable was the rate of reaction. When you changed the surface area it affected the rate of reaction. Everything else was controlled and kept contant. Nothing was overlooked, the experiment was flawless.
With the whole tablet it took about 7:50 min to completely dissolve in the water. As the tablet broke down more and more surface area was expose so the tablets reaction was sped up. This also happed to the tablet that was cut up. So when presented on a scale on how fast the reactions were the line would gradually go up because the rate of reactions would speed up. These are several things I did not predict. Another thing I found out was when the powered tablet was poured into the water it did not sink right away it floated at the top for about half the time before slowly sinking to the bottom once there it wasn’t very long before it had completely dissolved. This affected the experiment because when the powder is floating at the top it is not exposed to the water therefore the rate of reaction is slow and then when it sinks it suddenly speeds up. All data was constant and met up to what was predicted and more surface area was exposed the faster the rate of reaction became.
Overall the experiment was very successful. If I were to do this again I would make sure that the power sunk to bottom or perhaps use heavier tablet particles. No problems were found that disturbed the quality or the information or results were found. If I could do this project again I would try using different types of solutions that the tablet dissolved in to see which one was the fastest. This experiment was fun and I hope to do something like this again.
Conclusion
This experiment was testing the effect of surface area to the rate of reaction. All in all the experiment was a success, everything went as predicted. As I predicted the rate of reaction increased with more surface area exposed. The whole tablet took 7:50 min to dissolve; the cut tablet took 6:47 min dissolve and the crushed tablet took 1:02 min to dissolve. The experiment proved my hypothesis correct and was a joy to conduct. I continue to look forward to another experiment soon.

 RSS Feed
RSS Feed
